Learn
Delve into dermatology diseases with new approaches from conventional to complementary care.
Topics
Explore a broad spectrum of dermatological conditions and topics to enhance your practice.
Training Programs
Expert led instruction for every level of patient care.
Log in to see enrolled program content.
Conferences
Gain practical insights and meet new colleagues.
Hover over a conference to view recordings.
Join Us
The largest integrative dermatology community.
New and Emerging Topical Treatments for Eczema

The common eczema treatment regimen focuses on achieving four main goals: anti-inflammation, moisturization, anti-microbial, and anti-itch[1-3] (Figure 1). Anti-inflammatory therapy is commonly believed to be the pillar of eczema management. Nevertheless, these four approaches are typically implemented simultaneously to achieve maximum treatment benefits.

Figure 1. Treatment Goals
Credit: Vivian Shi
Over the past three decades, clinicians and scientists have made observations that have significantly increased our understanding of the causes of eczema and how our immune system interacts with our genes and environment to influence the disease.[4] Our immune system is made up of very complex pathways that communicate with each other through chemical signaling in order to achieve immune balance. In people with eczema, one or more segments of the immune system are not regulated properly, resulting in an overactive or dysregulated immune reaction causing skin inflammation. Some of the molecules that play a role in dysregulating immune pathways in people with eczema include interleukin-4, interleukin-13, interleukin-5, interleukin-33, interleukin-31, and phosphodiesterase-4. Several other molecules are currently being studied by researchers.
Since the 1970s, the incidence of eczema has increased dramatically, especially in developed countries.[5] However, development of new medications that target specific, deranged immune pathways in eczema (“targeted therapy”) has been limited. In fact, there has not been any new medication for eczema approved by the FDA for 15 years, between 2001 and 2016. Compared to psoriasis, another common inflammatory skin condition that has seen a dramatic increase in the number of new treatment options over the past 15 years, new eczema treatments have lagged behind. Medications approved for children with eczema are even more limited. In 2017, only two new medications were approved for eczema. Dupilumab is an injectable medication targeting the interleukin-4 and 13 pathways and Crisaborole is a topical ointment targeting the phosphodiesterase-4 pathway.[4] We are finally entering a new era for targeted eczema treatments. Currently, several other targeted eczema medications are under development and clinical investigation.
How Do Traditional Eczema Medications Differ From Targeted Treatments?
Our immune system can be thought of as an upside-down tree, with a tree trunk giving rise to large then small braches, and the leaves at the very far ends (Figure 2). In eczema, small segments of the immune function are abnormal; examples include the interleukin-4 and 13 pathways.[4] Traditional and targeted eczema medications both work by limiting the overactive immune function in our body and skin to lower skin inflammation. Traditional eczema medications suppress immune pathways higher up in the immune cascade (thought of as the tree trunk or large branches). On the other hand, targeted eczema medications suppress pathways much lower in the immune cascade (thought of as the small branches and leaves) to hone-in on the specific problems. Nevertheless, traditional medications are still effective in treating eczema and some people with eczema may have more than one dysregulated immune pathways and require broader immunosuppression.

Figure 2. Immune Cascade
Credit: Madsen S, Hendricks AJ, Shi VY*. “Growing Patient Education in Dermatology - An Upside-Down Tree Analogy for Immunomodulatory Therapies”. J Drugs Derm, 2020. PMID: 32227798.
Why Are Targeted Treatments Better?
When a relatively large portion of our immune function is suppressed, as in the case of traditional eczema treatments (non-targeted treatments), our ability to fight infections and certain cancers is weakened. Therefore, doctors may not want to keep their eczema patients on these medications for a prolong period of time.
What Are Some Potential Side Effects of Targeted Eczema Treatments?
Medication reactions will differ from person to person and medication to medication.
Targeted medications have shown favorable side effect profiles, but doctors and researchers will continue to learn about the benefits and potential side effects of these medications. Topical medications are usually the first line treatment when a person’s eczema is mild and affects limited body surface area. Systemic medications (taken by mouth, injection, or intravenously) are typically added when a person’s eczema is moderate to severe, involving a large body surface area, or when topical medications cannot adequately control the disease. Since the most common route for administering systemic targeted therapy is by injection, the most common side effect is injection site reactions, including variable degrees of redness, burning, and itching. The most common side effect of targeted topical treatment is burning and stinging upon application, and can be due to the active ingredients and/or other chemicals in the topical formulation.
Are Any Other Targeted Eczema Medications in the Pipeline?
Topically applied medications generally have less serious side effects and therefore receive FDA-approval more readily for use in children. Systemically administered medications are typically first approved for adults; approval in teenagers and school-aged children will likely take place over several years. Currently there is no clinical trial studying targeted eczema medications in children under 2 years old.
A few other new targeted eczema treatments are being developed and studied in clinical trials. Some of these target other interleukins, immune, and itch mediators associated with eczema. As researchers continue to decipher the intricate immune pathways involved in the development of eczema, new targeted treatments will become available. Future research will likely include studies looking at the benefit and safety of combination regimens, such as combining various traditional medications with targeted treatments, as well as combining one or more targeted treatments.
References
- Sidbury R, Tom WL, Bergman JN, et al. Guidelines of care for the management of atopic dermatitis: Section 4. Prevention of disease flares and use of adjunctive therapies and approaches. J Am Acad Dermatol.2014;71(6):https://pubmed.ncbi.nlm.nih.gov/25264237/.
- Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol.2014;https://pubmed.ncbi.nlm.nih.gov/24813302/.
- Sidbury R, Davis DM, Cohen DE, et al. Guidelines of care for the management of atopic dermatitis: section 3. Management and treatment with phototherapy and systemic agents. J Am Acad Dermatol.2014;https://pubmed.ncbi.nlm.nih.gov/24813298/.
- Malik K, Heitmiller KD, Czarnowicki T. An Update on the Pathophysiology of Atopic Dermatitis. Dermatol Clin.2017;https://pubmed.ncbi.nlm.nih.gov/28577801/.
- Silverberg JI. Public Health Burden and Epidemiology of Atopic Dermatitis. Dermatol Clin.2017;https://pubmed.ncbi.nlm.nih.gov/28577797/.
Related Articles

Atopic Dermatitis
Atopic Dermatitis Case Discussion with Dr. Peter Lio

Atopic Dermatitis
Navigating the Complexities of Atopic Dermatitis and Prurigo Nodularis: A Clinical Toolkit for Practitioners
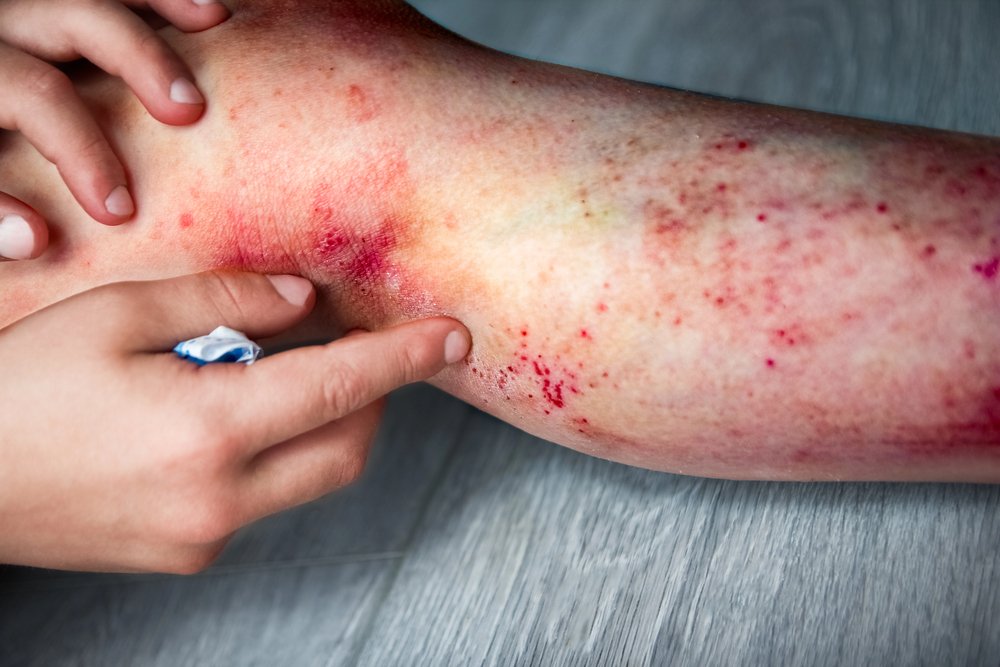
Atopic Dermatitis
Navigating Atopic Dermatitis and Hand & Foot Dermatitis



